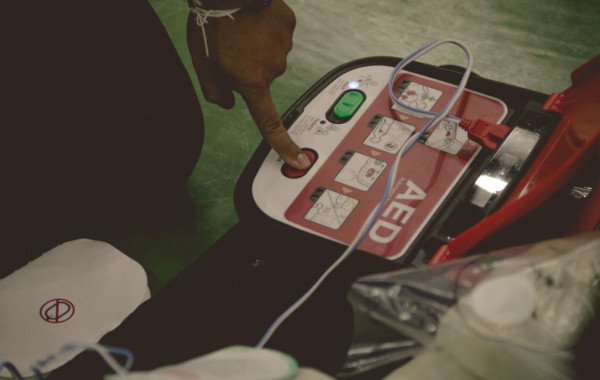
The Health Products Regulatory Authority (HPRA) is today stating that an estimated 583 automated external defibrillators (AEDs) require urgent safety updates in Ireland.
Without these updates, the cardiac devices across the five affected models may not work as intended in an emergency situation.
The devices' ongoing safety, and any updates which may be required, are monitored as part of the manufacturer’s post-market surveillance; they may require a software upgrade or the replacement of a component part within the defibrillator device.
The number of devices which require updates has dropped by 22 percent compared to the same period in 2016.
The HPRA is calling on all organisations and individuals in possession of an AED to check that their device is not one of the models affected.
This information may be found by looking at the dedicated AED webpage on the HPRA’s website which includes essential information on safety notices published by the manufacturers of affected devices, as well as details on how to contact the manufacturer directly to organise device updates.
The HPRA also has an information leaflet on AEDs available to download online with printed copies also available to order.
The 5 AED models which require corrective actions are:
- LIFEPAK 1000 defibrillator Physio-Control, Inc. USA
- Life-Point Metsis Medikal Teknik Sistemler Elektronik Otomoti
- Samaritan PAD 500P Physio-Control, Inc. USA
- Zoll AED Plus Zoll
- Samaritan Pad PAD 300, PAD 300P Physio-Control, Inc. USA
"This year, over 583 automated external defibrillators in Ireland require an urgent update, without which the devices may not perform in a life threatening emergency," Anne Tobin, the HPRA’s Medical Devices Vigilance Manager said.
"We know that the majority of cardiac arrests occur outside of the healthcare environment, where these devices offer an important first response intervention. We would urge those in possession of a device which needs to be upgraded to contact the manufacturer and to organise the necessary updates as soon as possible; this action could be the difference between life and death for whomever next requires treatment with the automated external defibrillator.
"As well as ensuring devices are updated as required, it is also so important to store and maintain automated external defibrillators as instructed by the manufacturer. During winter months, these devices may be affected by dropping temperatures and environmental conditions, so it is particularly important this time of year to ensure devices are stored appropriately."
In recent years, defibrillators have become easier to use, automatic, portable and affordable, which has resulted in a significant increase in the number of AEDs in Ireland.
Many Irish sporting venues, schools, hotels, restaurants, businesses and shopping centres now have the cardiac devices on their premises in case of emergencies.
All organisations with an AED on their premises should provide the manufacturer of their device with their correct contact details to ensure that the manufacturer can inform them of the need for safety upgrades if required.